Abstract
-
Purpose
This study investigated the possibility of ureteral stent removal in patients with gynecological cancer and ureteral obstruction.
-
Materials and Methods
From February 2010 to March 2022, the records of 121 patients with gynecological cancer and ureteral obstruction undergoing periodic ureteral stent replacement were reviewed retrospectively. Ureteral stent removal was attempted in patients who showed improvement in hydronephrosis on abdominal computed tomography (CT) after ureteral stent insertion and who lost symptoms. Success was defined as not having to reinsert the stent because there were no uncomfortable symptoms. The following were evaluated as predictors for ureteral stent removal: pre-removal serum creatinine, hypertension, diabetes, asymptomatic bacteriuria, type of gynecologic malignancy, prior radiotherapy, hydronephrosis laterality, obstruction level, hydronephrosis etiology, ureteral stent size, and ureteral stent duration. The hydronephrosis changes on abdominal CT were also evaluated.
-
Results
Among 121 patients, 74 with 81 obstructed ureters were evaluated. Of the 81 ureters, successful removal occurred in 30 (37.0%). There were significant differences between the success and failure cases regarding asymptomatic bacteriuria, prior radiotherapy, and hydronephrosis etiology. Logistic regression analysis showed that asymptomatic bacteriuria and hydronephrosis etiology were significant predictors of ureteral stent removal (p<0.05). There were also significant differences in the hydronephrosis changes depending on asymptomatic bacteriuria and hydronephrosis etiology (p<0.05).
-
Conclusions
In patients with gynecological cancer who have a ureteral stent in place due to ureteral obstruction, ureteral stent removal may be considered in patients who do not have asymptomatic bacteriuria or other etiologies, such as external ureteral pressure or ureteral cancer metastasis.
-
Keywords: Gynecology, Hydronephrosis, Bacteriuria, Stents, Neoplasms
INTRODUCTION
Gynecological cancer can cause chronic or, rarely, acute urinary tract obstruction in the pelvis or retroperitoneum [
1,
2]. The possible causes of urinary tract obstruction include direct metastasis of a malignant tumor or external pressure on the ureter by a tumor or malignant lymph node. In addition, adhesions in or around the ureter may occur because of surgery or radiation therapy. For cases of malignant ureteral obstruction, the insertion of a ureteral stent can be an effective treatment given appropriate patient selection based on renal function and tumor progression [
3].
Once inserted, ureteral stents are replaced periodically. Several studies have attempted to determine the predictors of stent failure in patients with malignant ureteral obstruction and a ureteral stent. Izumi et al. [
4] reported an unfavorable prognosis in patients with serum creatinine (Cr) of 1.2 mg/dl or higher and no gynecological treatment, such as chemotherapy or radiation after ureteral stent insertion. The authors also found gynecological cancer to be a good predictor of stent failure-free survival compared to other cancers. Wang et al. [
5] also reported that an Eastern Cooperative Oncology Group performance status of 2 or more, hydronephrosis >30 mm, and bladder involvement were independent predictors of failure. Maintaining a ureteral stent, even given a favorable prognosis, can be excruciating given the lifelong commitment. Surgical treatments, such as ureter end-to-end anastomosis or ureteroneocystomy, are options for patients wishing to avoid regular stent replacement.
Kurata et al. [
6] reported the clinical course of patients with indwelling ureteral stents due to hydronephrosis that developed during medical treatment for gynecological cancer. They reported that ureteral stent removal was possible in 18/45 (40.0%) ureters, all because of improved hydronephrosis, which occurred because the cancer or lower ureter obstruction compressed the ureter. On the other hand, this study did not determine the factors that might lead to recurrent hydronephrosis after ureteral stent removal. Therefore, this study examined which patients with gynecological cancer and ureteral obstruction were the best candidates for ureteral stent removal.
MATERIALS AND METHODS
This study received approval from the Institutional Review Board at CHA Bundang Medical Center (Protocol No. CHAMC2019-02-018). The records of 121 patients with gynecological cancer and ureteral obstruction who underwent periodic ureteral stent replacement from February 2010 to March 2022 were reviewed retrospectively. Among these patients, patients who underwent ureteral stent removal were analyzed. The inclusion criteria were as follows: (1) aged ≥18 years, (2) patients with gynecologic cancers (cervical, uterine, and ovarian cancer), (3) available abdominal computed tomography (CT) scans before and after ureteral stent removal, and (4) undergoing periodic ureteral stent replacement after radical hysterectomy. This study excluded patients who underwent urologic surgery that may affect hydronephrosis (e.g., ureterolithotomy and pyeloplasty), those whose renal dysfunction could affect urine production (estimated glomerular filtration rate <60 ml/min/1.73 m2), and those taking medications that could affect the ureter function (e.g., alpha-blockers, anti-cholinergics, and antihistamines). Ureteral stent removal was attempted in patients who showed improvement in hydronephrosis on abdominal CT after ureteral stent insertion and who did not show symptoms, such as flank pain. The patients underwent abdominal CT scans at least four weeks after ureteral stent removal. The ureteral stent was reinserted if hydronephrosis was noted on CT, same-side flank pain persisted, or symptoms, such as nausea, vomiting, or fever, occurred. Success was defined as cases without stent reinsertion because there were no uncomfortable symptoms after ureteral stent removal.
Hydronephrosis was defined as an increase in the anterior-posterior (AP) diameter of the renal pelvis on abdominal CT. The AP diameter was measured in the axial plane, where the renal pelvis leaves the kidney [
7]. The AP diameter was measured three times on the abdominal CT (e.g., before ureteral stent insertion, after ureteral stent insertion, and at least four weeks after ureteral stent removal), and these measures were used to determine the change in AP diameter.
The following were evaluated: patientʼs age, body mass index (BMI), history of diabetes or hypertension, and whether radiation therapy was received after a radical hysterectomy for gynecological cancer. Urinalysis and urine culture were performed intermittently during the follow-up period after ureteral stent insertion, and results obtained at least four weeks before ureteral stent removal were evaluated. Urine samples were collected using the clean catch technique, and bacteriuria was defined as cases involving bacterial growth of ≥105 colony-forming units (CFU)/ml. Asymptomatic bacteriuria was defined as the isolation of bacteria from a urine culture without urinary tract infection symptoms. Hydronephrosis laterality (right, left, and bilateral) and level (upper, middle, and lower) were investigated on abdominal CT before ureteral stent insertion. The hydronephrosis etiologies were also evaluated on abdominal CT (e.g., external ureter pressure from the tumor or abdominal lymph nodes, ureter metastasis, or periureteral infiltration). External ureter pressure was defined as a case of a confirmed tumor mass or lymph node enlargement outside the ureter that compresses the ureter. Ureter metastasis was defined as focal circumferential thickening of the ureter with transmural involvement of the ureteral wall. Periureteral infiltration was defined as focal thickening of the ureter that may be associated with ill-defined extra-luminal soft tissue. The ureteral stent size and the time from the initial ureteral stent insertion to removal were also investigated.
Statistical analyses were performed using SPSS 19.0 software (IBM Co., Armonk, NY, USA). The differences in the variables between patients with successful and failed ureteral stent removal were tested using a Student t-test and chi-squared test. Univariate and multivariate logistic regression analyses were performed to identify the significant factors that predict successful ureteral stent removal. The regression analysis results are presented as odds ratios (ORs) and 95% confidence intervals (CIs). One-way repeated-measures analysis of the variance (ANOVA) was used to evaluate the changes in AP diameter measured on abdominal CT scans. The effect size for statistical analysis was ηp2. A Greenhouse–Geisser correction was used when the Mauchlyʼs test of sphericity was violated. Post hoc pairwise comparisons with a Bonferroni adjustment were applied when significant interaction effects were observed. A p-value <0.05 was considered significant.
RESULTS
Among the 121 patients assessed initially, 74 patients with 81 obstructed ureters meeting the inclusion criteria (81 ureters: a unilateral ureter from 67 patients and a bilateral ureter from 7 patients) were evaluated. The average age was 53 years (33-74 years), and the average duration of ureteral stent maintenance before ureteral stent removal was 26 months (9-59 months). The urine culture revealed a positive result in 35 (43.2%) of the 81 cases; asymptomatic bacteriuria was confirmed because there were no accompanying symptoms. Among the 35 cases, Escherichia coli was identified in 14 cases (40.0%), Enterococcus spp. in eight cases (22.9%), Klebsiella spp. in seven cases (20.0%), and Pseudomonas aeruginosa in six cases (17.1%) as the cause for asymptomatic bacteriuria.
Among the gynecological cancer patients, cervical cancer (n=39 ureters) was the most common, followed by uterine cancer (n=26 ureters) and ovarian cancer (n=16 ureters,
Table 1). Radiation therapy was administered to 39 (48.1%) ureters. The most common obstruction level was the lower ureter in 63 cases (77.8%). Abdominal CT showed that the external pressure of the ureter and cancer metastasis were possible etiologies in 23 and 29 ureters, respectively. Periureteral infiltration was observed in 29 ureters. During ureteral stent removal, seven out of 74 patients survived with no evidence of disease after gynecological treatment, while 67 survived with disease and were receiving gynecological treatment.
Ureteral stent removal was successful in 30 (37.0%) of the 81 ureters. Significant differences were found between success and failure after ureteral stent removal in patients with a ureteral obstruction in terms of asymptomatic bacteriuria (p=0.002), prior radiotherapy (p=0.041), and hydronephrosis etiology (p=0.001) (
Table 1). The patientsʼ age, BMI, pre-removal serum Cr, duration of ureteral stent placement, hypertension, diabetes, type of gynecological cancer, hydronephrosis laterality, obstruction level, and ureteral stent size did not differ between patients with successful and failed ureteral stent removal.
Table 2 lists the results of univariate and multivariate logistic regression analyses of the clinical factors as predictors of successful ureteral stent removal. Multivariate logistic regression analyses showed that asymptomatic bacteriuria (OR, 0.277; 95% CI, 0.079-0.970; p=0.045) and hydronephrosis etiology were significant predictors of successful ureteral stent removal (external pressure [OR, 0.231; 95% CI, 0.062-0.863; p=0.029] and ureter invasion [OR, 0.116; 95% CI, 0.026-0.520; p=0.005]). Multivariate analyses revealed prior radiotherapy as not predictive of successful ureteral stent removal.
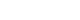
One-way repeated-measure ANOVA was conducted to compare the changes in AP diameter measured on abdominal CT (
Fig. 1). The difference in the AP diameter change was significant depending on asymptomatic bacteriuria (F [2, 158]=10.554, p=0.001, η
p2=0.118,
Fig. 1A). Furthermore, the difference in AP diameter change was significant depending on the hydronephrosis etiology (F [4, 156]=3.142, p=0.016, η
p2=0.075,
Fig. 1B).
DISCUSSION
Most studies have reported factors that predict ureteral stent failure in patients with malignant ureteral obstruction [
3-
5]. Once inserted, the stent requires regular replacement for the remainder of the patientʼs life or until stent failure. Although replacing a ureteral stent may not be a major issue for patients undergoing cancer treatment, a periodic replacement can be burdensome. Sometimes patients inquire if ureteral stent removal is possible. On the other hand, few studies have examined the likelihood of successful stent removal in patients with a ureteral obstruction. This study examined the factors related to successful ureteral stent removal in patients undergoing periodic ureteral stent replacement.
Ureteral stent removal was successful in 30 (37.0%) of the 81 ureters. The presence of asymptomatic bacteriuria and a hydronephrosis etiology emerged as significant predictors of successful stent removal in patients with a ureteral obstruction (
Table 2). In addition, this study further investigated whether the presence of asymptomatic bacteriuria and hydronephrosis etiology affected the AP diameter following AP diameter changes after stent removal. Although AP diameter increased gradually after stent removal in most ureters, the changes in AP diameter were significantly different over time depending on the presence of asymptomatic bacteriuria and hydronephrosis etiologies, as confirmed by CT (
Fig. 1).
After ureteral stent removal, the AP diameter increased over time, secondary to external ureter pressure from a tumor, abdominal lymph nodes, or metastasis. On the other hand, slight increases were observed secondary to periureteral infiltration. Ureteral obstruction is accom-panied by periureteral infiltration because ureteral cancer develops inside the ureter [
8]. However, in cases of gynecological cancer, periureteral infiltration does not always cause ureter obstruction because the tumor is generated externally. In some cases, the degree of ureteral obstruction is less than that of an external tumor, lymph node pressure, or cancer metastasis to the ureter itself. These results were also confirmed in the present study.
The effects of asymptomatic bacteriuria on successful ureteral stent removal are unknown. It is generally accepted that urinary stasis allows bacteria to attach to the urothelium, thereby increasing the risk of infection [
9,
10]. Because patients with ureteral obstruction were included in the study, asymptomatic bacteriuria is more likely to cause discomfort after ureteral stent removal by increasing the risk of infection. Eventually, these patients are more likely to have a ureteral stent reinsertion, as shown in this study. This study identified
E. coli as the most common cause of asymptomatic bacteriuria, followed by
Enterococcus spp.,
Klebsiella spp., and
P. aeruginosa. Although there were some differences between the reported studies,
Staphylococcus spp.,
E. coli,
Klebsiella spp.,
P. aeruginosa,
Enterococcus spp., and
Candida spp. were most often responsible for ureteral stent colonization [
11]. Unfortunately, few studies have reported the treatment of confirmed asymptomatic bacteriuria when considering ureteral stent removal.
The serum Cr before ureteral stent removal and prior radiotherapy would determine the success or failure after stent removal. Serum Cr ≥1.2 mg/dl before ureteral stenting was reported as predictive of stent failure [
4,
12]. There are conflicting reports on the effect of radiotherapy on the ureter. Yeaman et al. [
13] reported that ureteral strictures were significantly more common in patients who received radiation than those who did not. On the other hand, Horan et al. [
14] reported that radiotherapy did not cause a deterioration of the renal function or hydronephrosis severity among patients with stage IIIB cervical cancer. In the present study, serum Cr or radiotherapy exposure was not associated with successful ureteral stent removal, and there was no significant difference in the AP diameter changes relative to these factors.
The present study had several limitations. First, the disease progression and treatment results of gynecological cancer were not considered when analyzing the success of stent removal. A more successful treatment of gynecological cancer would increase the possibility of successful ureteral stent removal. On the other hand, this retrospective study analyzed stent removal in patients who showed improvement in hydronephrosis on abdominal CT after ureteral stent insertion and who did not show symptoms, such as flank pain during gynecological cancer treatment and who did not reflect the disease progression and treatment results of gynecological cancer. Furthermore, these results are expected to be helpful when considering the removal of a ureteral stent because of complaints of discomfort during gynecological treatment, as most of the patients included in the study were under similar conditions and receiving chemotherapy after gynecological cancer surgery.
Second, the observation period after ureteral stent removal through medical record analysis was short, generally within three months, because patients who could be followed up for at least four weeks after the ureteral stent removal and had abdominal CT results were included in the study. Nevertheless, cases of ureteral stent reinsertion may increase if the follow-up period becomes longer. In addition, although the ureteral stent was removed due to the discomfort of the patients, there may be patients who need to maintain the ureteral stent or consider urinary diversion, such as percutaneous nephrostomy, because preservation of renal function is a priority. Therefore, long-term follow-up after ureteral stent removal will be necessary for future studies.
Confirming asymptomatic bacteriuria may differ because a urine culture is not performed immediately before ureteral stent removal. Nevertheless, the urine culture results may not significantly affect the results because they were usually used within one month before ureteral stent removal.
Another limitation concerns the histological condition of periureteral infiltration confirmed by abdominal CT. Höckel et al. [
15] examined patients with ureteral obstruction secondary to local cervicovaginal cancer. They reported that a malignant ureteral obstruction might result in (1) secondary to tumor infiltration by associated retroperitoneal fibrosis of the mesometrium, (2) malignant infiltration of the mesureter, or (3) infiltration of the ureter muscle. In this study, periureteral infiltration on abdominal CT indicated tumor infiltration by the associated retroperitoneal fibrosis of the mesometrium or malignant infiltration of the mesureter. On the other hand, it is not always easy to distinguish the above-mentioned histopathological findings accurately with abdominal CT.
Finally, because this was a retrospective study, instruments, such as the Ureteric Stent Symptoms Questionnaire, were not used to collect information on the stent-related quality of life and symptoms [
16,
17]. This study investigated ureteral stent reinsertion to determine the success or failure of ureteral stent removal. Nevertheless, some patients may dislike reinserting the ureteral stent, despite some discomfort after the ureteral stent removal. Using these questionnaires during ureteral stent insertion and maintenance will allow an objective investigation of the discomfort it causes the patient. This analysis can provide considerable information on ureteral stent reinsertion into the patient. Future studies on patients with gynecological cancer and ureteral obstruction undergoing periodic ureteral stent replacement should consider these limitations.
CONCLUSIONS
In patients with gynecological cancer and ureteral obstruction undergoing periodic ureteral stent replacement, the reinsertion of a ureteral stent will likely occur after stent removal if there is asymptomatic bacteriuria or other etiologies, such as external ureteral pressure or ureteral cancer metastasis. These should be considered before removing the ureteral stent.
NOTES
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
S.H.J. participated in data collection and wrote the manuscript. Y.K.H., Y.D.Y., and S.R.L. participated in the study design and performed the statistical analysis. S.R.L. participated in the study design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
Fig. 1Changes in the anterior-posterior (AP) diameter measured on abdominal computed tomography scans. (A) Change in the AP diameter according to the hydronephrosis etiology. (B) Changes in the AP diameter according to asymptomatic bacteriuria. Pre-stent: AP diameter measured before ureteral stent insertion, Post-stent: AP diameter measured after ureteral stent insertion, Stent removal: AP diameter measured at least four weeks after ureteral stent removal.
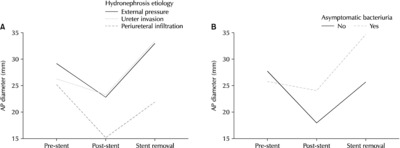
Table 1Comparison of the variables between success and failure after ureteral stent removal
|
Variables |
|
Number of ureters |
|
Total (n=81) |
Success (n=30) |
Failure (n=51) |
p-value*
|
|
Age (y) |
|
53.4±9.6 |
53.9±9.2 |
53.2±9.9 |
0.740a)
|
|
BMI (kg/m2)
|
|
24.2±3.7 |
23.5±3.8 |
24.7±3.6 |
0.173a)
|
|
Serum Cr (mg/dl)c)
|
|
0.9±0.4 |
0.8±0.4 |
0.9±0.4 |
0.554a)
|
|
Ureteral stent duration (mo) |
|
26.3±16.3 |
25.4±17.8 |
26.8±15.6 |
0.718a)
|
|
Hypertension |
No |
44 |
19 |
25 |
0.212b)
|
|
Yes |
37 |
11 |
26 |
|
|
Diabetes |
No |
61 |
22 |
39 |
0.752b)
|
|
Yes |
20 |
8 |
12 |
|
|
Asymptomatic bacteriuria |
No |
46 |
25 |
21 |
0.002b)
|
|
Yes |
35 |
5 |
30 |
|
|
Gynecologic malignancy |
Cervical |
39 |
19 |
20 |
0.055b)
|
|
Uterine |
26 |
5 |
21 |
|
|
Ovarian |
16 |
6 |
10 |
|
|
Prior radiotherapy |
No |
42 |
20 |
22 |
0.041b)
|
|
Yes |
39 |
10 |
29 |
|
|
Hydronephrosis laterality |
Right |
37 |
17 |
20 |
0.235b)
|
|
Left |
30 |
10 |
20 |
|
|
Bilateral |
14 |
3 |
11 |
|
|
Obstruction level |
Upper |
9 |
3 |
6 |
0.934b)
|
|
Middle |
9 |
3 |
6 |
|
|
Lower |
63 |
24 |
39 |
|
|
Hydronephrosis etiology |
External pressure |
23 |
7 |
16 |
0.001b)
|
|
Invasion of ureter |
29 |
4 |
25 |
|
|
Periureteral infiltration |
29 |
19 |
10 |
|
|
Ureteral stent size (Fr.) |
<6 |
19 |
7 |
12 |
0.984b)
|
|
≥6 |
62 |
23 |
39 |
|
Table 2Logistic regression analysis for the factors affecting successful ureteral stent removal
|
Parameter |
Univariate |
Multivariate |
|
p-value |
OR |
95% CI |
p-value |
OR |
95% CI |
|
Asymptomatic bacteriuria |
0.002 |
0.14 |
0.046-0.425 |
0.045 |
0.277 |
0.079-0.970 |
|
Prior radiotherapy |
0.043 |
0.379 |
0.148-0.971 |
0.341 |
0.575 |
0.184-1.796 |
|
Periureteral infiltrationa)
|
|
1 |
|
|
1 |
|
|
External pressure |
0.002 |
0.143 |
0.042-0.490 |
0.029 |
0.231 |
0.062-0.863 |
|
Invasion of ureter |
0.001 |
0.057 |
0.014-0.222 |
0.005 |
0.116 |
0.026-0.520 |
REFERENCES
- 1. Ganatra AM, Loughlin KR. The management of malignant ureteral obstruction treated with ureteral stents. J Urol 2005;174:2125-8. ArticlePubMed
- 2. Sountoulides P, Pardalidis N, Sofikitis N. Endourologic management of malignant ureteral obstruction: indications, results, and quality-of-life issues. J Endourol 2010;24:129-42. ArticlePubMed
- 3. Chung SY, Stein RJ, Landsittel D, Davies BJ, Cuellar DC, Hrebinko RL, et al. 15-Year experience with the management of extrinsic ureteral obstruction with indwelling ureteral stents. J Urol 2004;172:592-5. ArticlePubMed
- 4. Izumi K, Mizokami A, Maeda Y, Koh E, Namiki M. Current outcome of patients with ureteral stents for the management of malignant ureteral obstruction. J Urol 2011;185:556-61. ArticlePubMed
- 5. Wang JY, Zhang HL, Zhu Y, Qin XJ, Dai BO, Ye DW. Predicting the failure of retrograde ureteral stent insertion for managing malignant ureteral obstruction in outpatients. Oncol Lett 2016;11:879-83. ArticlePubMed
- 6. Kurata S, Tobu S, Udo K, Noguchi M. Outcomes of ureteral stent placement for hydronephrosis in patients with gyne-cological malignancies. Curr Urol 2017;10:126-31. ArticlePubMedPMCPDF
- 7. Glodny B, Rapf K, Unterholzner V, Rehder P, Hofmann KJ, Strasak A, et al. Accessory or additional renal arteries show no relevant effects on the width of the upper urinary tract: a 64-slice multidetector CT study in 1072 patients with 2132 kidneys. Br J Radiol 2011;84:145-52. ArticlePubMedPMC
- 8. An T, Kwon GY, Min JH, Park SY. Ureteral cancer: complete ureteral obstruction on computed tomography urography is associated with periureteral fat invasion. Eur J Radiol 2020;130:109198. ArticlePubMed
- 9. Cox CE, Hinman F Jr. Experiments with induced bacteriuria, vesical emptying and bacterial growth on the mechanism of bladder defense to infection. J Urol 1961;86:739-48. ArticlePubMed
- 10. Heyns CF. Urinary tract infection associated with conditions causing urinary tract obstruction and stasis, excluding uroli-thiasis and neuropathic bladder. World J Urol 2012;30:77-83. ArticlePubMedPDF
- 11. Bey E, Bouiller K, le Goux C, Pimpie R, Tourret-Arnaud J, et al. Vallée M. Epidemiology and risk factors for ureteral stent-associa-ted urinary tract infections in non-transplanted renal patients: a systematic review of the literature. World J Urol 2021;39:3845-60. ArticlePubMedPDF
- 12. Yu SH, Ryu JG, Jeong SH, Hwang EC, Jang WS, Hwang IS, et al. Predicting factors for stent failure-free survival in patients with a malignant ureteral obstruction managed with ureteral stents. Korean J Urol 2013;54:316-21. ArticlePubMedPMC
- 13. Yeaman CT, Winkelman A, Maciolek K, Tuong M, Nelson P, Morris C, et al. Impact of radiation on the incidence and management of ureteroenteric strictures: a contemporary single center analysis. BMC Urol 2021;21:101. ArticlePubMedPMCPDF
- 14. Horan G, McArdle O, Martin J, Collins CD, Faul C. Pelvic radiotherapy in patients with hydronephrosis in stage IIIB cancer of the cervix: renal effects and the optimal timing for urinary diversion? Gynecol Oncol 2006;101:441-4. ArticlePubMed
- 15. Wolf B, Hentschel B, Horn LC. Höckel M. Surgical treatment and histopathological assessment of advanced cervicovaginal carcinoma: a prospective study and retrospective analysis. Eur J Cancer 2017;70:99-110. ArticlePubMed
- 16. Joshi HB, Newns N, Stainthorpe A, MacDonagh RP, Keeley FX Jr, Timoney AG. Ureteral stent symptom questionnaire: develop-ment and validation of a multidimensional quality of life measure. J Urol 2003;169:1060-4. ArticlePubMed
- 17. Joshi HB, Stainthorpe A, Keeley FX Jr, MacDonagh R, Timoney AG. Indwelling ureteral stents: evaluation of quality of life to aid outcome analysis. J Endourol 2001;15:151-4. PubMed
 , Young Kwon Hong
, Young Kwon Hong , Young Dong Yu
, Young Dong Yu , Seung-Ryeol Lee,
, Seung-Ryeol Lee,

 , Department of Urology, CHA Bundang Medical Center, CHA University School of Medicine, 59, Yatap-ro, Bundang-gu, Seongnam 13496, Korea, Tel: +82-31-780-5350, Fax: +82-31-780-5323, E-mail: seung102@cha.ac.kr
, Department of Urology, CHA Bundang Medical Center, CHA University School of Medicine, 59, Yatap-ro, Bundang-gu, Seongnam 13496, Korea, Tel: +82-31-780-5350, Fax: +82-31-780-5323, E-mail: seung102@cha.ac.kr 


 KAUTII
KAUTII
 ePub Link
ePub Link Cite
Cite